|
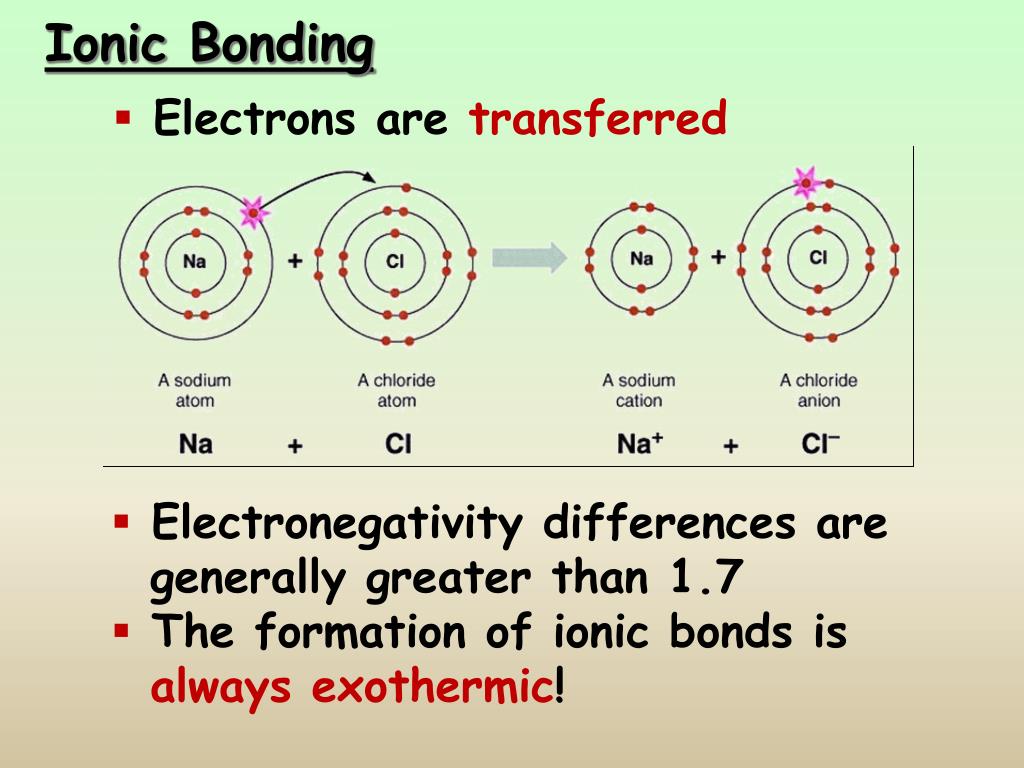
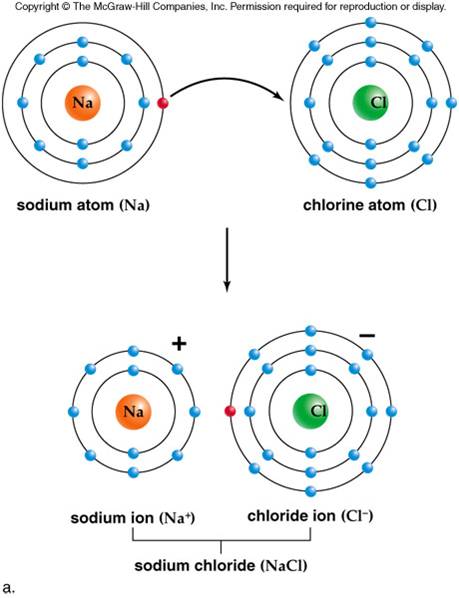
The presence of such paths was later indicated by, e.g., AFM analyses by Frischat et al. Transport properties and the relation to glass composition are mostly understood on the basis of conductivity/diffusion paths across which the considered ions are transported, as originally proposed by Greaves (1985). The glass composition has received much attention over the years and the specific components of alumina ( Burggraaf, 1966), phosphate ( Bartholomew and Garfinkel, 1980), and zirconia ( Nordberg et al., 1964) have been shown to have beneficial effects on chemical strengthening through acting on free volume, oxygen packing density, and degree of cross-linking of the structural backbone. In the present understanding of ion-exchange kinetics, the transport coefficient is governed by many different parameters, such as salt-bath composition (the diffusion reservoir), glass composition (the structure and free volume of the matrix), exchanging ion species (size and charge), temperature, pressure, etc. Where in a simplistic approximation, D 1 and D 2 are the coefficients of self-diffusion of contributing species 1 and 2, N 1 and N 2 are the corresponding relative atomic fractions, and D ¯ is the effective diffusion coefficient. The transport coefficient is usually concentration-dependent ( Doremus, 1964), While usually, the concentration gradient (∂c/∂x) is considered, the actual driving force for ionic transport is the gradient in chemical potential (∂μ/∂x) between the different ionic media ( Schaeffer, 2012) (which in many cases, correlates linearly with the gradient in chemical composition). The ion-exchange process can be described as an interdiffusion reaction where the rate of reaction is described by Fick’s laws of diffusion ( Nordberg et al., 1964). Monovalent cation exchange has also received much attention for tailoring the refractive index profile of the surface layer, i.e., so as to create microstructured planar or buried waveguides or optical lenses with graded refractive index ( Ramaswamy and Srivastava, 1988 Opilski et al., 2000 Honkanen et al., 2006 Tervonen et al., 2011). This specific subject has been developing into a large market, where ion exchange strengthened glasses are now used in displays, handheld electronic devices, pharmaceutical packaging, and many other areas. In particular, alkali cation exchange has received significant attention throughout the years, primarily with respect to introducing compressive stresses into the surface layer, which increases mechanical durability ( Gy, 2003 Karlsson et al., 2010a Varshneya, 2010a, b). Numerous properties can be modified in this way, including mechanical, optical, electrical, and chemical performance. In this context, ion exchange has long been known as a useful tool to modify the surface of glass products ( Schulze, 1913 Stern, 1966). This enables highly effective ion-exchange processes. Monovalent cations exhibit pronounced mobility in silicate glasses. Correlations of effective diffusion coefficients were found for the bond dissociation energy and the electronic cation polarizability, indicating that localization and rupture of bonds are of importance for the ion-exchange rate. In the present paper, we discuss the trends in the effective diffusion coefficients when exchanging Na + for various monovalent cations (K +, Cu +, Ag +, Rb +, and Cs +) by drawing relations to physicochemical properties. Basic understanding of the relations between structural parameters and the effective diffusion coefficients may help to improve ion-exchanged SLS glass products, on the one hand in terms of obtainable strength and on the other in terms of cost. This largely excludes the major area of soda-lime-silicates (SLS) such as are commodity in almost all large-scale applications of glasses. However, most of the present applications rely on specifically tailored matrix compositions in which the cation mobility is enhanced. In particular, alkali cation exchange has received significant attention, primarily with respect to introducing compressive stress into the surface region of a glass, which increases mechanical durability. Numerous properties can be modified in this way, e.g., mechanical, optical, electrical, or chemical performance. Monovalent cations enable efficient ion-exchange processes due to their high mobility in silicate glasses.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
